Step-by-step explanation:
Molarity is defined as the number of moles divided by one liter of solution.
Mathematically, Molarity =
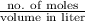
As it is given that volume is 2.0 L and mass is 10.0 grams. Therefore, first we will calculate the number of moles as follows.
No. of moles =
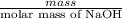
=
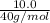 (molar mass of NaOH = 40 g/mol)
(molar mass of NaOH = 40 g/mol)
= 0.25 mol
Hence, calculate the molarity of the solution as follows.
Molarity =
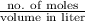
=
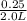
= 0.125 M
Thus, we can conclude that molarity of given solution is 0.125 M.