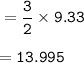Moles of H₂ are needed to produce 9.33 moles of NH₃ : 13.995
Further explanation
A reaction coefficient is a number in the chemical formula of a substance involved in the reaction equation. The reaction coefficient is useful for equalizing reagents and products.
The reaction coefficient in a chemical equation shows the mole ratio of the reactants and products
Reaction for the synthesis of ammonia :
N₂+3H₂⇒2NH₃
moles of NH₃ = 9.33
From equation, mol ratio of H₂ : NH₃ = 3 : 2, so mol H₂ :