Answer:
597K
Step-by-step explanation:
Given parameters:
Initial pressure = 1.2atm
Initial temperature = 125°C = 125 + 273 = 398K
Initial volume = 1.00L
Final pressure = 1.80atm
Final volume = 1.00L
Unknown:
Final temperature = ?
Solution:
To solve this problem, we must understand that at constant volume, the pressure of a given mass of gas varies directly with the absolute temperature.
Mathematically;
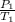 =
=
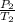
where P and T are the pressures
1 and 2 are the initial and final states
Now insert the parameters and solve;
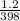 =
=
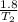
T₂ x 1.2 = 398 x 1.8
T₂ = 597K