Step-by-step explanation:
Photoelectric effect: Electrons are ejected out from the surface of metal when light of sufficient frequency falls upon shiny surface of metals. light is made up small bundles of energy packets named photons which when falls on surface transfer their energy to electrons due which electrons ejects out of the metallic surface. The energy for one photon was given by expression:
E=h
 , or
, or
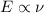
h=Planck's constant,
 = frequency of the light
= frequency of the light
Kumar can increase the energy of emitted electron by using light with higher value of frequency than the frequency of red light. This is because the energy carried by the photon of a light is directly proportional to the frequency of the light. Higher the value of frequency of light higher will be the value of energy of a photon. And photon with higher energy will impart more amount of energy to an electron while ejection.