Answer: Two of them are balanced and two of them are unbalanced.
Step-by-step explanation:
A balanced chemical equation is defined as the equation in which total number of individual atoms on the reactant side is equal to the total number of individual atoms on the product side. These equations follow law of conservation of mass.
An unbalanced chemical equation is defined as the equation in which total number of individual atoms on the reactant side is not equal to the total number of individual atoms on the product side. These equations does not follow law of conservation of mass.
For the given chemical equations:
Equation 1:

As, number of carbon, nitrogen and oxygen atoms on the reactant side is equal to the number of carbon, nitrogen and oxygen atoms on the product side. Thus, this is a balanced chemical equation.
Equation 2:
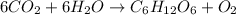
As, number of oxygen atoms on the reactant side is not equal to the number of oxygen atoms on the product side. Thus, this is not a balanced chemical equation.
Equation 3:
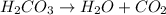
As, number of carbon, nitrogen and oxygen atoms on the reactant side is equal to the number of carbon, nitrogen and oxygen atoms on the product side. Thus, this is a balanced chemical equation.
Equation 4:
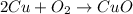
As, number of oxygen and copper atoms on the reactant side is not equal to the number of oxygen and copper atoms on the product side. Thus, this is not a balanced chemical equation
Hence, two of the equations are balanced and two are unbalanced.