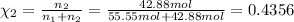Step-by-step explanation:
1) Mass of carbon dioxide = 100 g
Molar mass of carbon dioxide = 44 g/mol
Moles of carbon dioxide =
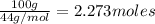
1 mol = 0.001 kmol
2.273 moles= 2.273 × 0.001 kmol =
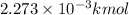
2) 1 liter of ethyl alcohol of density
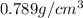
Volume of ethyl alcohol ,V= 1 L = 1000 mL
Density of ethyl alcohol =d =
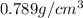
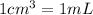
Mass of ethyl alcohol = m
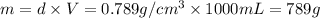
Molar mass of ethyl alcohol = 46 g/mol
Moles of ethyl alcohol =
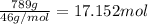
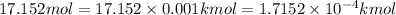
3) Volume of oxygen gas,V =
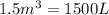
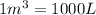
Temperature of the gas = T= 25°C = 298.15 K
Pressure of the gas ,P= 1 atm
Moles of oxygen gas = n
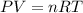
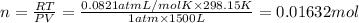
0.01632 mol = 0.01632 × 0.001 kmol=
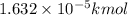
4) Volume water in mixture = 1 L
Density of water =
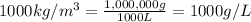
Mass of water =
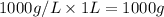
Volume of alcohol = 2.5 L
Density of alcohol =
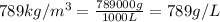
Mass of alcohol =
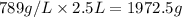
Mass of mixture = 1000 g + 1972.5 g = 2972.5 g
Mass percentage of water :
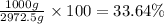
Mass percentage of alcohol :
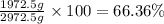
Moles of water :
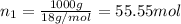
Moles of alcohol =
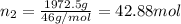
Mole fraction of water :
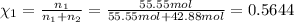
Mole fraction of alcohol :