Answer:
The mole to mole ratio between oxygen gas and water is 2 : 1
Step-by-step explanation:
Balancing is making the number of atoms of each element same on both the sides (reactant and product side).
To find the number of atoms of each element we multiply coefficient and the subscript
For example
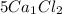 contains
contains
5 × 1 = 5 ,Ca atoms and
5 × 2 = 10, Cl atoms
If there is a bracket in the chemical formula
For example
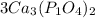
we multiply coefficient × subscript × number outside the bracket to find the number of atoms
(Please note: 3 is the coefficient, and if there is no number given then 1 will be the coefficient )
So
3 × 3 = 9 , Ca atoms
3 × 1 × 2 = 6, P atoms
3 × 4 × 2 = 24, O atoms are present.
So
The balanced chemical equation is
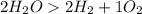
The number which stands in front of the chemical formula is the coefficient and it represents the number of moles of each substance.
So we see here 2 is the coefficient of
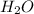 and 1 is the coefficient of
and 1 is the coefficient of
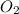
So the mole ratio of water and oxygen gas is 2 : 1
(Answer)