Answer: The mass of gallium produced by the electrolysis is 0.0354 grams.
Step-by-step explanation:
The equation for the deposition of Ga(s) from Ga(III) solution follows:
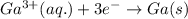
- To calculate the total charge, we use the equation:
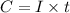
where,
C = charge
I = current = 0.490 A
t = time required (in seconds) =
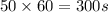 (Conversion factor: 1 min = 60 s)
(Conversion factor: 1 min = 60 s)
Putting values in above equation, we get:
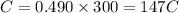
- To calculate the moles of electrons, we use the equation:
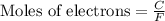
where,
C = charge = 147 C
F = Faradays constant = 96500
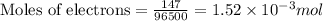
- Now, to calculate the moles of gallium, we use the equation:
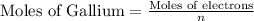
where,
n = number of electrons transferred = 3
Putting values in above equation, we get:
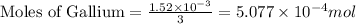
- To calculate the mass of gallium, we use the equation:
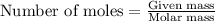
Moles of Gallium =
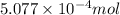
Molar mass of Gallium = 69.72 g/mol
Putting values in above equation, we get:
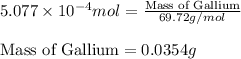
Hence, the mass of gallium produced by the electrolysis is 0.0354 grams.