Answer:
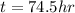
Step-by-step explanation:
Hello!
In this case, given the mass of aluminum metal which can undergo the following reduction from aluminum oxide:
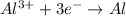
We can see three moles of electrons are transferred per mole of aluminum, thus, we obtain:
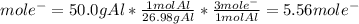
Next, we compute the charge carried by those electrons via the faraday's constant:
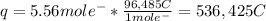
Finally, considering the applied current, we compute the elapsed time in hours to achieve such electrolysis:
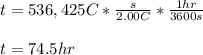
Best regards!